Means take as needed1/25/2024 
In 2011, the FDA added a Risk Evaluation and Mitigation Strategy (REMS) to the dispensing requirements for mifepristone permitting only medical providers who had obtained certification from the manufacturer to prescribe and directly dispense the drug. Risk Evaluation and Mitigation Strategies (REMS) telehealth organizations have been providing the misoprostol-only regimen as an option for m edication a b or t i on fo r a n umber of years. When taken, the misoprostol-only regimen successfully t erminates the pregnancy approximately 80-100 % of the time, with a complication rate of less than 1%. Research has shown the misoprostol-only regimen to be a safe and highly effective method of pregnancy termination, however it may result in a higher incidence of side effects, particularly diarrhea, fever and chills. It involves taking 800 µg (4 pills) of misoprostol sublingually or vaginally every three hours for a total of 12 pills. The regimen is also recommended for up to 70 days (10 weeks) of pr egnancy. While the combined regimen of mifepristone and misoprostol for medication abortion is recommended, there is a second medication abortion protocol using mis o prostol – only that is more commonly used internationally and currently not approved by the FDA. 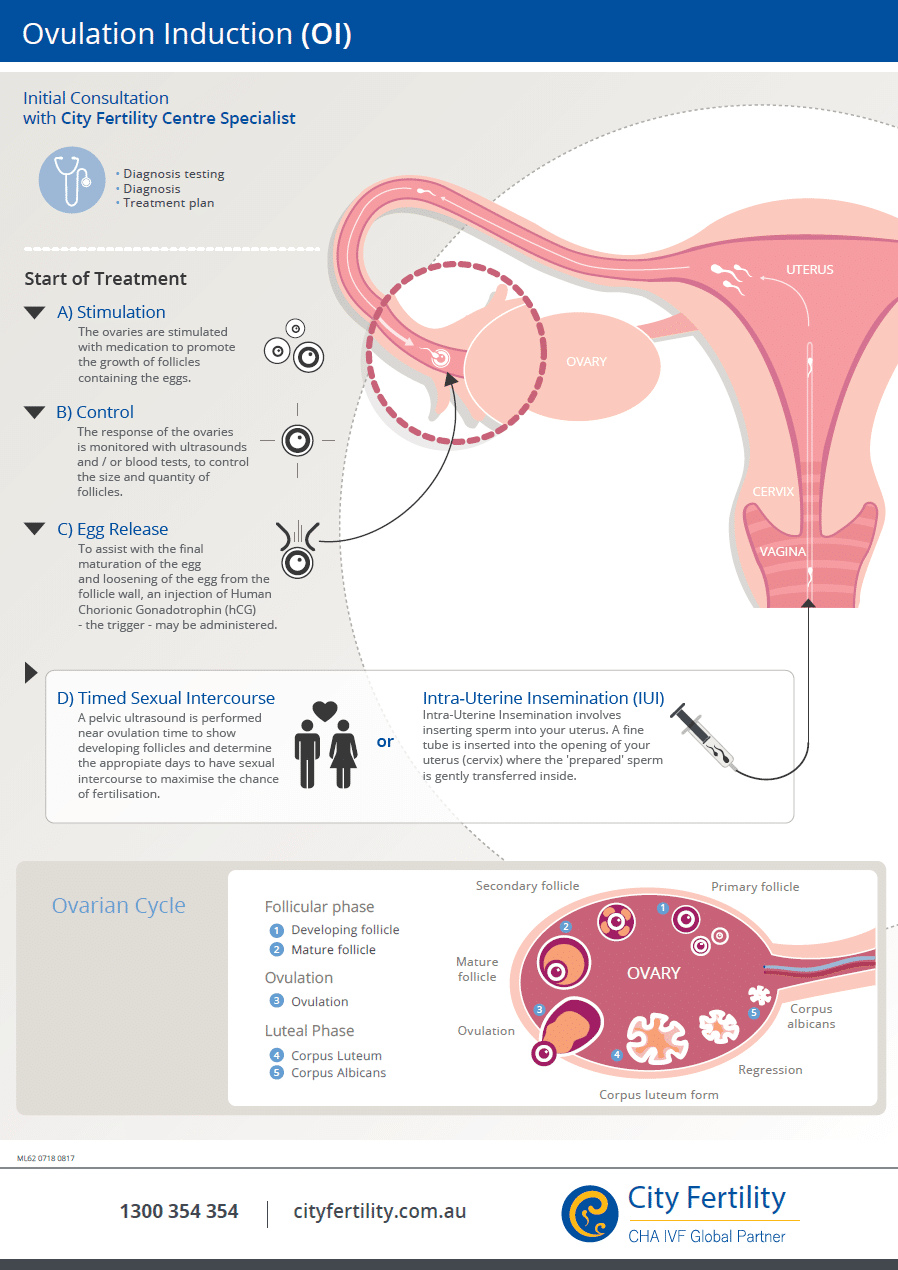
In 2019, the FDA approved GenBioPro, Inc.’s application for generic mifepristone. Until 2019, mifepristone was only sold under the brand name Mifeprex, manufactured by Danco Laboratories. 
This regimen approves use of medical abortions for up to 70 days (10 weeks) of pregnancy ( Table 1). In 2016, the FDA updated and approved a new evidence-based regimen and drug label, which guides current clinical practice. When taken, medication abortion successfully terminates the pregnancy 99.6% of the time, with a 0.4% risk of major complications, and an associated mortality rate of less than 0.001 percent (0.00064%). The FDA has found that medication abortion is a safe and highly effective method of pregnancy termination. A follow-up visit can be scheduled a week or two later, to confirm that the pregnancy was terminated via ultrasound or blood test. Misoprostol, taken 24–48 hours after mifepristone, works to empty the uterus by causing cramping and bleeding, similar to an early miscarriage. Mifepristone works by blocking progesterone, a hormone essential to the development of a pregnancy, and thereby preventing an existing pregnancy from progressing. Mifepristone, also known as the abortion pill, or RU-486 is sold under the brand name Mifeprex and through a generic manufactured by GenBioPro in the United States. The most common medication abortion regimen in the United States involves the use of two different medications: mifepristone and misoprostol. Both involve taking oral medicat ions to t erminate a pregnancy. There are two medication abortion regimens that have a long safety and efficacy record : m ifepristone with m isoprostol and misoprostol alone. This factsheet provides an overview of medication abortion, how it is used and regulated, the role of the drug in self-managed abortions, and an analysis of the intersection of federal and state regulations pertaining to its provision and coverage. 
The medication abortion drug regimen approved by the FDA is available in many states across the nation, however, dispensing these pills for the purpose of terminating a pregnancy is now banned in some states. By 2021, over half of abortions in the US were medication abortions. Since the FDA first approved the drug in 2000, its use in the United States has quickly grown. The World Health Organization has authorized use to 12 weeks of pregnancy. Medication abortion, also known as medical abortion or abortion with pills, is a pregnancy termination protocol that involves taking two different drugs, Mifepristone and misoprostol, that can be safely used up to the first 70 days (10 weeks) of pregnancy according to the U.S. This has created a new focus on medication abortion as an option for expanding access to people facing barriers to abortion care. States can now set their own policies protecting or banning abortion without any federal standard protecting access to abortion. Jackson Women’s Health Organization, overturning Roe v. On June 24 th, 2022, the Supreme Court ruled on the Dobbs v. Note: Figure 1 and Table 1 were updated on Apto reflect recent changes in several states.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
